Oil- and water-based formulations were frequently used for cosmetic and
dermatological treatments or as process chemicals. It is well known that
the specific behaviour concerning the dermal penetration and irritation
potential will be modified during and after the course of application
due to supersaturated solutions by evaporization of water and other volatile
components (1). It can also be influenced by ingredients of which the
distribution coefficient is not known before.
The aim of this in-vitro skin study was to learn the dermal behaviour of newly developed ferrofluides (nanoparticles) dispersed in water- and oil-based formulations under different application conditions.
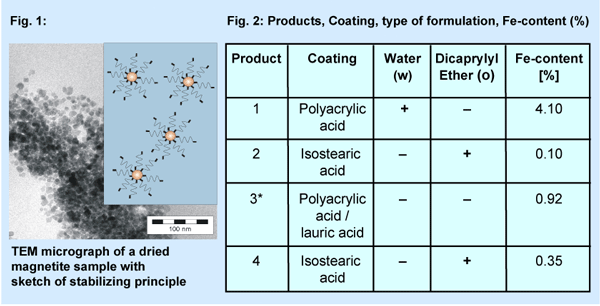
Ferrofluids are nanoparticles either magnetic or non-magnetic dis-persed in solvent (Fig. 1, Fig. 2). Two water-based products (product 1, 3) contain magnetite-particles coated by polyacrylic acid and lauric acid (product 3*) respectively. The other two samples (magnetite or zinc-ferrit) are based on the emollient dicaprylyl ether as vehicle and isostearic acid (Cognis Deutschland) as coating material. The product 4 contains zinc-ferrit instead of magnetite.
To achieve long-term stability in water and organic solvents respectively, the particle size has been adjusted to 5 - 20 nm and the particle surface has been modified to adapt the polarity of the particles to the dispersing agent. The core of the nanoparticles consists of non-toxic iron oxide and was synthesised by precipitation of ferric salt precursors. A suspension of these particles is long-term stable especially by applying a magnetic field. The surface modification consists of fatty acids which shows a good adhesion to iron oxide.
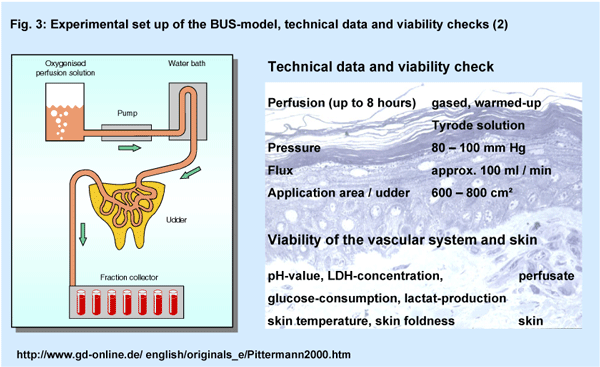
The isolated perfused bovine udder skin (BUS) is regarded as a viable
in-vitro model (Fig. 3). Using unimpaired natural
skin it takes into account active skin barrier properties and cutaneous
metabolic processes, so that it be used to study for both, penetration
and irritation induced by ingredients or formulations (2, 3, 4, 5).
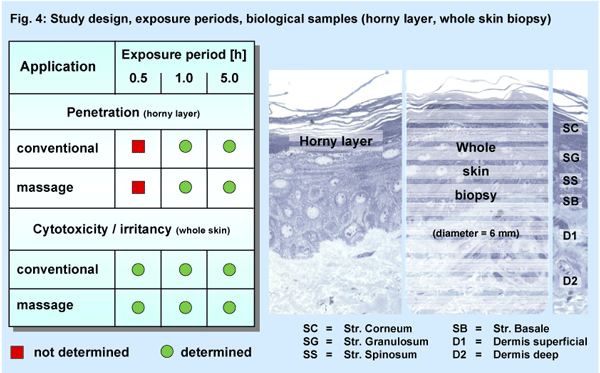
The open application (2 - 4 g / 100 cm2) was performed in four independent studies (Fig. 4) either by the means of glass spatula (conventional) or a intensive finger massage (1 minute) of the skin. After the exposure periods of 1.0 h and 5.0 h the repeated horny layer stripping method (10 times) was used (Tesa 4204, BDF, Hamburg, 19 mm × 100 mm) for studying the penetration of Fe. For the irritation assays a short term exposure period of 0.5 h was added to the periods of 1.0 h and 5.0 h. Whole skin biopsies were prepared for the MTT-assay (cytotoxicity) and the determination of the PGE2-tissue concentration (irritancy) compared to untreated sites.
Additionally a new technique for a precise histochemical determination
of six different layers of the skin was introduced (Fig.
4). In contrast to the standard preparation of the whole skin biopsies
this procedure (V-max-technique, HistoServe GmbH, Münster, Germany)
allows to assay the activities of certain intracellular enzymes in freely
selected regions of epidermal or dermal layers.
On the basis of the permanent viability of the BUS-model the time related
activation or recovery of the natural skin can be analysed and compared
after topical applications or manipulation e.g. skin massage.
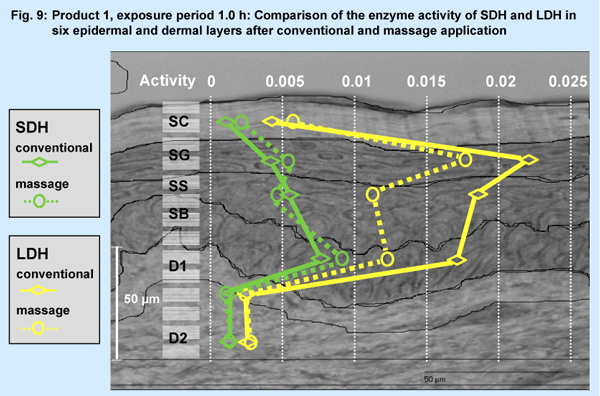
A selected result (Fig. 9; product 1, conventional vs. massage application,
exposure period 1.0 h) is presented for LDH (lactate-dehydrogenase, in
SG mainly) and SDH (succinate-dehydrogenase, in SB mainly).
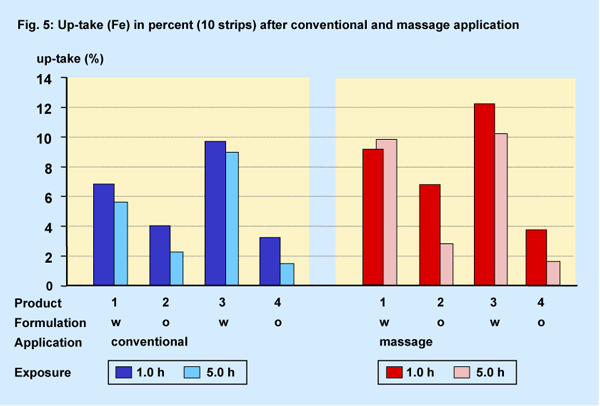
Penetration Calculated up-take (%)
Due to the various Fe-content in the products a calculation of the actual
up-take (percentage) into the horny layer was made on the assumption that
a fixed amount of product (2 g / 100 cm2) was applied.
The percentual up-take was at the maximum approx. 10 % - 12 % (Fig.
5). As expected the 1st strips of all groups exhibited more Fe-content
(> 1.0 %) than each of the following nine strips at both exposure periods.
Regarding the influence of the time period of exposure only the oil-based
formulations (o) showed a significant decrease of Fe analysed in the horny
layer after the prolonged exposure period.
A clear product differentiation between water- and oil-based formulations
(w, o) was also calculated under massage conditions for the exposure period
of 1.0 h. Higher amounts of Fe compared to conventional application were
analysed after the short exposure period for all products except the product
4.
After the long exposure period the Fe-content of the water-based formulations
remains about on a similar level. So the massage effect regarding the
calculated up-take was not generally relevant even after the prolonged
exposure period.
Irritation (cytotoxicity / irritancy)
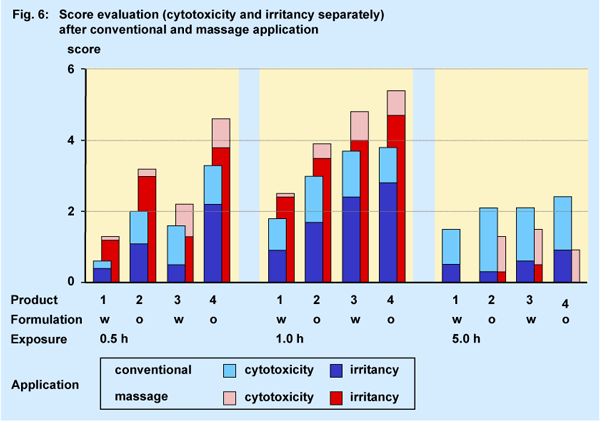
For the irritation assay a very short exposure period of 0.5 h was included into the study design additionally. The degree of irritation is demonstrated by the score value combined from the relative MTT-result (cytoxicity, irreversible) and the PGE2-concentration (irritancy, reversible) (6, 7). All columns in Fig. 6 consist of both, the singular score for irritancy on the basis and for the cytotoxicity on the top.
Only after the very short exposure period of 0.5 h a difference between
the water- or oil-based formulations could be observed. The score value
of the oil-based formulations (product 2, 4) was more than twofold compared
to the value of the water-based formulations.
At the exposure period of 1.0 h no distinction between the different formulations
was observable any more due to the delayed increase of the score value
of the water-based products. Each formulation displayed its own specific
score profile unrelated to its individual Fe-content and type of formulation.
No product differentiation could be also observed after the prolonged
exposure period of 5.0 h. The score values were significantly diminished
on the basis of the almost complete reversibility of the preinflammatory
mediator prostaglandin E2-concentration.
As expected intensive massage application induces an increased score level compared to the conventional application due the intensified release of PGE2. Under massage conditions the change from the formulation-related profile at the exposure period of 0.5 h to the individual product profile after additional 30 minutes was very similar to the results obtained after the conventional application.
Generally the massage procedure also altered the relationship between cytotoxic and irritant action of all products independently from the type. The mechanical skin treatment by the massage induced an increase of the PGE2-tissue concentration by about 70 %, whereas simultaneously the cytotoxic activity of the formulations was decreased by approx. 50 %.
Although the tape stripping did not result in the complete removal of
all of the stratum corneum and contents of the hair follicles the material
recovered should be treated as representative fraction of that present
in the horny layer due to the extra large size of the strips.
But regarding the size of the nanoparticles (5 - 20 nm) a certain amount
of ferrofluids could assumed to be remained within the acro-infundibulum
of the hair follicles (Fig. 7).
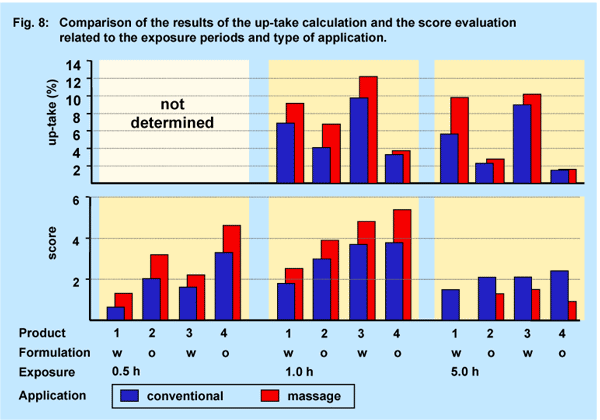
The results shown in the Figures 5 and 6 are combined in Fig. 8 for direct comparison. Clearly the penetration potential (up-take) into the horny layer is differently profiled than the irritation potential (score).
The results (Fig. 9, relative units) of the V-max-technique showed no altered activity for the enzyme SDH with its main activity in SB. However, the massage treatment induced a slight decrease of activity of LDH in SB, SS and SG, but not in the horny and dermal layers.
- The Fe-content and the type of formulation did influence the results at the short term exposure period up to 1.0 h. After prolonged exposure periods later on unspecific reactions regarding the skin irritation capability were induced. The dermal behaviour regarding the penetration capability remains unchanged even after 5.0 h exposure.
- Ferrofluids used as nanoparticles do not interfere with the specific type of formulation. The actual Fe-content and the massage effect do not influence the irritation potential specifically.
- The results regarding the dermal behavior of the ferrofluids indicates a high skin compatibility even after prolonged exposure periods and intensive massage during application. No abrasive effects on the skin could be detected.
- Using the V-max-technique alterations of intracellular enzyme activities (SDH, LDH) within six epidermal and dermal layers related to the type of application could be differentiated.SDH remained unchanged even after massage application.
1. Th. Förster, W. Pittermann, M. Schmitt and M. Kietzmann; Skin
penetration properties of cosmetic formulations using a perfused bovine
udder model; J. Cosmet. Sci. 50, 147-157 (1999)
2. M. Kietzmann, W. Löscher, D. Arens, P. Maaß and D. Lubach;
The Isolated Perfused Bovine Udder as an in Vitro Model of Percutaneous
Drug Absorption. Skin Viability and Percutaneous Absorption of Dexamethasone,
Benzoyl Peroxide and Etofenamate; J. Pharm.Toxicol. Meth. 30, 75-84 (1993)
3. W. Pittermann, B. Jackwerth and M. Schmitt (1997); The Isolated Perfused
Bovine Udder Skin Model: In Vitro Model for the Assessment of Skin Penetration
& Irritation; In Vitro Toxicol. 10, 17-21 (1/1997)
4. W. Bäumer and M. Kietzmann; The isolated Perfused Bovine Udder
as a Model of Dermal Eicosanoid Releaser; ATLA 28,123-134 (2000)
5. Ch. Hammes, M. Schmitt, Th. Förster and W. Pittermann; "Bioaktive
Wirkstoffe - Nachweis der Penetration" in Innovative Analytik in
der Kosmetik; 14. DGK-Symposium; Proceedings 91-93 (2001).
6. Nicotera, P.; Alteration of Cell Signalling in Chemical Toxicity; Arch.
Toxicol. (suppl. 18) pp 3-11; Springer Verlag, Berlin, Heidelberg (1996)
7. Ponec, M.: In vitro models to predict Skin Irritation in The Irritant
Contact Dermatitis Syndrom (edited by P.G.M. van der Valk, H.I.Maibach)
pp 335-341; CRC Press, Boca Raton (1995)